The bodys first line of defense versus SARS-CoV-2– before the participation of antibodies and T cells– is thought to depend upon receptor molecules such as RIG-I that recognize the viruss hereditary product and cause the production of signaling proteins referred to as type I interferons. These interferons, in turn, promote the production of proteins that can prevent viral duplication and promote the recruitment of immune cells to battle the infection.
Several studies have suggested that early and robust production of interferons secures against COVID-19, whereas delayed production is connected with severe disease. A clinical trial has revealed that dealing with COVID-19 patients with purified interferon protein early throughout disease can decrease mortality, however producing interferons is extremely costly.
In the new JEM research study, Iwasaki and coworkers recommend a cheaper alternative: treating patients with short RNA particles that imitate SARS-CoV-2s hereditary material and trigger the RIG-I receptor to stimulate production of type I interferons by the bodys own cells.
The researchers evaluated their technique in mice susceptible to SARS-CoV-2 infection. A single dose of an RNA particle named SLR14 was adequate to safeguard the mice from severe illness and death, particularly if the treatment was offered soon before or not long after direct exposure to the virus. When administered shortly after viral infection, SLR14 was more reliable than treating mice with cleansed interferon protein, the researchers identified.
Most importantly, SLR14 secured mice from all emerging SARS-CoV-2 versions, including the delta version responsible for the current surge in United States COVID-19 cases.
Iwasaki and coworkers checked SLR14 in immunocompromised Rag–/– mice chronically contaminated with SARS-CoV-2. The RNA particle was able to completely clear the infection from these animals, despite the fact that they do not have both T cells and antibody-producing B cells.
Iwasaki and coworkers explain that RNA particles like SLR14 are easy and relatively inexpensive to make. “SLR14 for that reason holds great promise as a new class of RNA therapies that can be applied as antivirals versus SARS-CoV-2,” Iwasaki states. “Moreover, since this RNA-based healing approach is flexible and simple, our study will help with pandemic preparedness and action versus future respiratory pathogens conscious type I interferons.”
Reference:” A stem-loop RNA RIG-I agonist secures versus persistent and acute SARS-CoV-2 infection in mice” by Tianyang Mao, Benjamin Israelow, Carolina Lucas, Chantal B.F. Vogels, Maria Luisa Gomez-Calvo, Olga Fedorova, Mallery I. Breban, Bridget L. Menasche, Huiping Dong, Melissa Linehan, Yale SARS-CoV-2 Genome Surveillance Initiative, Craig B. Wilen, Marie L. Landry, Nathan D. Grubaugh, Anna M. Pyle and Akiko Iwasaki, 10 November 2021, Journal of Experimental Medicine (JEM). DOI: 10.1084/ jem.20211818.
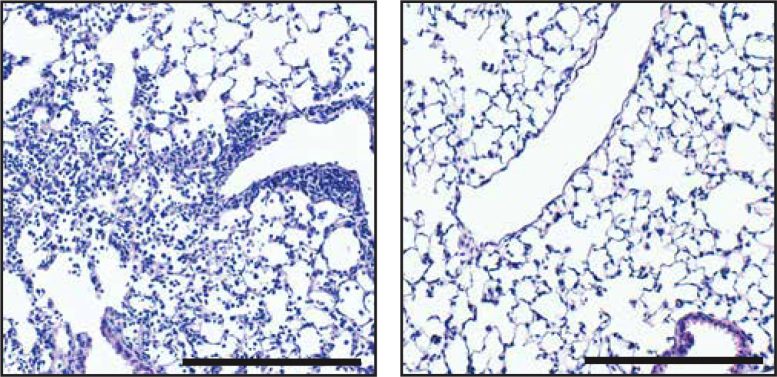
Compared to a control (left), treatment with SLR14 (right) lowers the signs of swelling and pneumonia in the lungs of SARS-CoV-2– infected mice. Credit: © 2021 Mao et al. Originally released in Journal of Experimental Medicine. https://doi.org/10.1084/jem.20211818
Scientists at Yale School of Medicine have discovered that an RNA molecule that promotes the bodys early antiviral defense system can protect mice from a variety of emerging SARS-CoV-2 versions. The research study, published recently in the Journal of Experimental Medicine (JEM), could lead to new treatments for COVID-19 in immunocompromised patients, as well as supplying an economical restorative option for establishing nations that presently do not have access to vaccines.
Though approved vaccines versus SARS-CoV-2 are highly effective at preventing extreme disease and death during the continuous COVID-19 pandemic, vaccine accessibility is extremely limited in numerous low-income countries, and brand-new, vaccine-resistant pressures of the virus might emerge in the future. Furthermore, the vaccines effectiveness is already lowered in immunocompromised individuals not able to form adequate varieties of antibodies or T cells that specifically target the viral spike protein. These people are prone to chronic, long-lasting SARS-CoV-2 infections.
” This is why, in addition to using vaccines in avoiding COVID-19, efforts are required to establish efficacious rehabs against SARS-CoV-2,” says Akiko Iwasaki, a professor at Yale School of Medicine.
Compared with a control (left), treatment with SLR14 (right) lowers the signs of inflammation and pneumonia in the lungs of SARS-CoV-2– contaminated mice. Authorized vaccines versus SARS-CoV-2 are highly reliable at avoiding severe disease and death during the continuous COVID-19 pandemic, vaccine accessibility is very restricted in numerous low-income nations, and new, vaccine-resistant stress of the infection might emerge in the future. The researchers evaluated their method in mice susceptible to SARS-CoV-2 infection. “SLR14 for that reason holds excellent pledge as a brand-new class of RNA therapies that can be used as antivirals against SARS-CoV-2,” Iwasaki states.